Abstract
Background : Aim of this study is to see the efficiency of
topical 1% hydrocortisone in the treatment of physiological
phimosis.
Materials and Methods: Retrospective study was
conducted in the Kaski Model Hospital, Pokhara, Gandaki,
Nepal from January 2016 to June 2019. Forty-nine patients
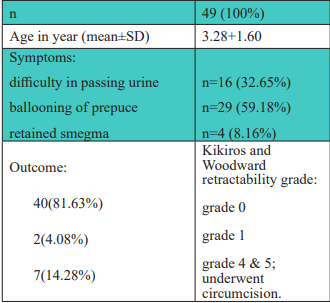
treated in outpatient basis have been selected for this study.
Selection was made according to Kikiros and Woodward
retractability grading.
Results: Out of 49(100%) patients 40(81.63%)
achieved complete response to steroid, which is
Kikiros and Woodward retractability grade 0. 2(4.08%)
patients showed satisfactory response (symptom
free) which is Kikiros and Woodward retractability
grade 1. And 7(14.28%) patients showed no response
after six weeks course of treatment and underwent
circumcision.
Conclusion: This study demonstrates the use of 1%
topical hydrocortisone is safe, simple and cost effective.
And it could be the initial choice of treatment.
References
Urology.2012; 2012:707329.
2. Ko MC, Lui CK, Lee WK, Jeng HS, Chiang
HS, Li CY. Age-specific prevalence rates of
phimosis and circumcision in Taiwanese boys.
J Formos Med Assoc. 2007;106(4):302-307.
3. Nascimento FJ, Pereira RF, Silva JL, Tavares
A, Pompeo AC. Topical Betamethasone and
Hyaluronidase in the Treatment of Phimosis in
Boys: a Double-Blind, Randomized,Placebo
controlled Trial. Int Braz J Urol. 2011 MayJun;37(3):314-319.
4. P. A. Dewan, H. C. Tieu, and B. S. Chieng,
“Phimosis: is circumcision necessary?”
Journal of Paediatrics and Child
Health.1996;32(4):285–289.
5. Ashfield JE, Nickel KR, Siemens DR,
Physiological Phimosis and The Use of Topical Steroid. Shrestha D. et. al.
~224~
Original Article Medical Journal of Pokhara Academy of Health Sciences Vol. 3 Issue 1
MacNeily AE, Nickel JC. Treatment of
phimosis with topical steroids in 194 children.
JUrol. 2003;169:1106-8.
6. Kuehhas FE, Miernik A, Sevcenco S, Tosev
G, Weibl P, Schoenthaler M, et al. Predictive
power of objectivation of phimosis grade
on outcomes of topical 0.1% betamethasone
treatment of phimosis. Urology. 2012;80:412-
6.
7. Moreno G, Corbalan J, Penaloza B, Pantoja T.
Topical corticosteroids for treating phimosis
in boys (Review).Cochrane Database of
Systematic Reviews. 2014; Issue 9. Art. No.:
CD008973.
8. Nobre YD, freitas RG, Felizardo MJ, Ortiz
V, et al. to cric or not to cric: clinical and
pharmoeconomic outcomes of a prospective
trial of topical steroid versus primary
circumsion. Int Braz J Urol. 2010;36(1):75-85
9. McGregor T, Pike J, Leonard M. Pathologic
and physiologic phimosis Approach to the
phimotic foreskin. Can Fam Physician.
2007;53:445-448.
10. Kikiros CS, Beasley SW, Woodward AA.
The response of phimosis to local steroid
application. Pediatr Surg Int. 1993;8:329-332.
11. Marques TC, Sampaio FJ, Favorito LA.
Treatment of phimosis with topical steroids
and foreskin anatomy. International Brazilian
Journal of Urology. 2005;31(4):370–4.
MEDLINE: 16137407
12. Zampieri N, Corroppolo M, Zuin V, Bianchi S,
Camoglio FS. Phimosis and topical steroids:
new clinical findings. Pediatric Surgery
International. 2007;23(4):331–5. MEDLINE:
17308904
13. J. M. Elmore, L. A. Baker, W. T. Snodgrass,
and P. Dewan, “Topical steroid therapy as
an alternative to circumcision for phimosis
in boys younger than 3 years,” Journal of
Urology. 2002;168(4):1746–1747.